
The sight of North Korean workers in the Russian Federation has historically been limited to the shadowed confines of Siberian logging camps or the skeletal frames of half-finished construction sites in the Far East. However, a profound transformation is currently underway as Moscow begins to

The landscape of modern oncology is shifting toward highly personalized treatment pathways, necessitating diagnostic precision that can only be achieved through the integration of sophisticated molecular and cellular analysis tools. Agilent Technologies has underscored its commitment to this

The rapid integration of biometric identification into the modern office environment has reached a critical threshold where convenience meets the absolute boundary of physical human autonomy. Lawmakers in Washington have responded to this technological encroachment by introducing House Bill 2303, a
The terrifying reality of catastrophic internal bleeding is that patients often succumb to their injuries before they can even reach the sterile environment of a modern operating room. In a landmark achievement for medical technology, Arsenal Medical has recently reported the first successful human
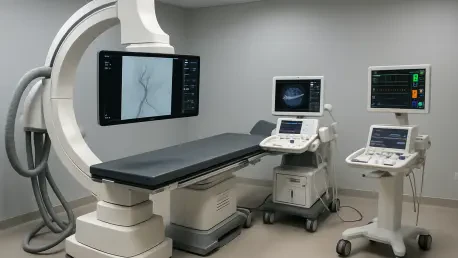
The delivery of sophisticated medical technology to the High-Specialty Regional Hospital of the Yucatán Peninsula in Mérida represents a transformative shift in the region's ability to provide life-saving care to its most vulnerable populations. Governor Joaquín Díaz Mena spearheaded this

The global healthcare market in 2026 continues to undergo a profound transformation as the intersection of an aging demographic and rapid technological evolution creates both unprecedented opportunities and significant operational pressures for medical device manufacturers. Analysts monitoring the