
Navigating the complexities of a rare diagnosis often requires far more than a simple prescription delivery; it demands a comprehensive support system that bridges the geographical and clinical gaps inherent in modern medicine. This article explores how specialized pharmacy models provide a

The landscape of American healthcare is currently undergoing its most significant structural shift since the inception of the Medicare program, as the federal government officially moves to tether technological innovation to measurable patient success. By implementing the Advancing Chronic Care
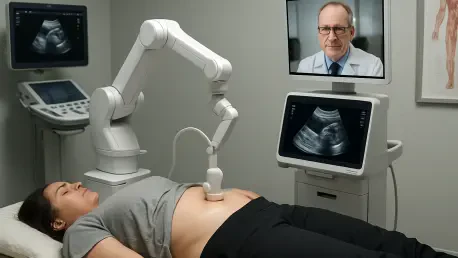
The ability to perform a life-saving medical procedure from a continent away has finally moved from the realm of speculative fiction into a tangible clinical reality for the global medical community. The geographic isolation of extreme environments often means that specialized medical diagnostics

The silent progression of high cholesterol remains one of the most significant yet underaddressed threats to cardiovascular longevity in the modern healthcare landscape. Statistics indicate that approximately 70% of adults struggling with obesity also contend with elevated cholesterol levels,
The rapid integration of artificial intelligence into clinical environments has transitioned from a theoretical possibility to a fundamental necessity for modern healthcare institutions aiming to provide precision medicine. Weill Cornell Medicine recently launched its institution-wide initiative,

The U.S. Food and Drug Administration has initiated a fundamental transformation of its regulatory oversight for digital health, ushering in an era where many low-risk technologies no longer require stringent premarket review. This strategic deregulation, detailed in guidance issued this year, is