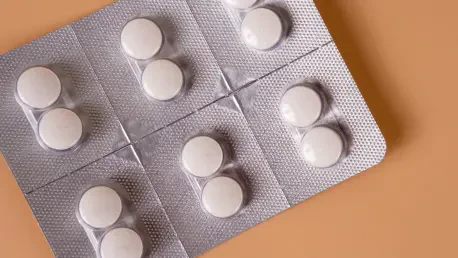
The landscape of reproductive healthcare in the United States faces another profound shift as the Supreme Court, through a decisive order issued by Justice Samuel Alito, temporarily reinstates broad access to the primary pharmaceutical option for terminating pregnancies. This specific intervention effectively pauses a previous attempt by a lower court to impose stringent regulations that would have significantly curtailed the availability of mifepristone across the nation. By granting this temporary stay, the justices have ensured that patients can continue to utilize modern delivery methods, including telehealth consultations and mail-order pharmacy services, which have become essential pillars of care. In the current climate of 2026, these medication-based procedures account for the vast majority of reproductive health interventions, serving as a critical resource for individuals in regions with limited surgical options. The ruling prevents an immediate disruption of services, allowing local pharmacies to operate without requiring the burdensome in-person doctor visits that were previously mandated by more restrictive judicial interpretations.
Legal Jurisdictions and the Regulatory Divide
The ongoing legal conflict surrounding the distribution of these medications highlights a deepening rift between federal pharmaceutical standards and individual state mandates. Several jurisdictions, led by officials in states like Louisiana, argue that the widespread availability of the pill through digital and postal channels bypasses local statutes designed to regulate medical practice. Conversely, many Democratic-led states have established robust legal shields for healthcare providers, creating a complex patchwork of protection and restriction that varies wildly across state lines. This friction has forced the Supreme Court to intervene while it reviews emergency appeals submitted by pharmaceutical manufacturers who seek to protect their FDA-approved distribution models. This one-week reprieve serves as a cooling-off period during which the high court can assess the broader implications of judicial interference in established medical protocols. While anti-abortion groups characterize the current distribution system as a form of unregulated trafficking, many health experts view these legal challenges as a direct threat to the scientific autonomy of federal regulators who oversee the safety and efficacy of modern medicine.
Strategic Adaptations for Healthcare Infrastructure
Healthcare providers and advocacy groups moved quickly to adjust their operational strategies during this period of extreme legal volatility. When the threat of more restrictive regulations first appeared, many clinics were forced to consider alternative protocols, such as switching to a less effective single-drug regimen to ensure continuity of care. However, the temporary restoration of access allowed these organizations to resume standard practices while preparing for various judicial outcomes. Moving forward, the focus shifted toward building more resilient healthcare networks that could withstand sudden shifts in the regulatory environment. Legal experts recommended that providers diversify their supply chains and strengthen their digital privacy protections to safeguard patient information in an increasingly scrutinized landscape. The commitment to maintaining access persisted despite the uncertainty of the high court’s final deliberations, as medical professionals prioritized the safety of their patients over the shifting political tides. By documenting the effectiveness of telehealth models, advocates successfully built a data-driven case for the permanent preservation of pharmaceutical autonomy.