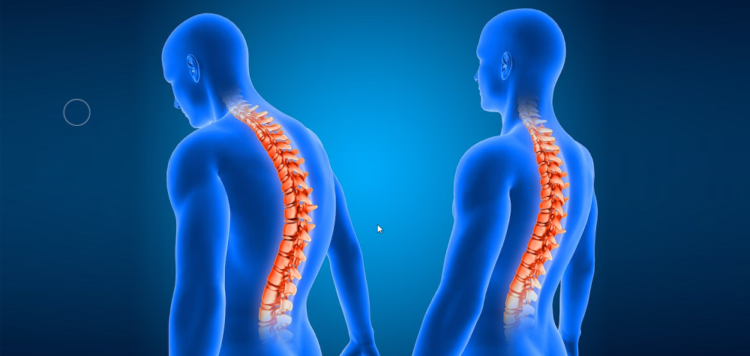
The US Food and Drug Administration (FDA) has granted approval for Globus Medical’s REFLECT Scoliosis Correction System.
Claimed to be the company’s first humanitarian device, REFLECT is intended for the correction of progressive scoliosis in young patients and preserve motion, while maintaining stability and enabling future modulated growth.
This system is designed for skeletally immature patients with progressive idiopathic scoliosis who need surgery to achieve and maintain correction of the spine.